How does ATP affect pyruvate dehydrogenase
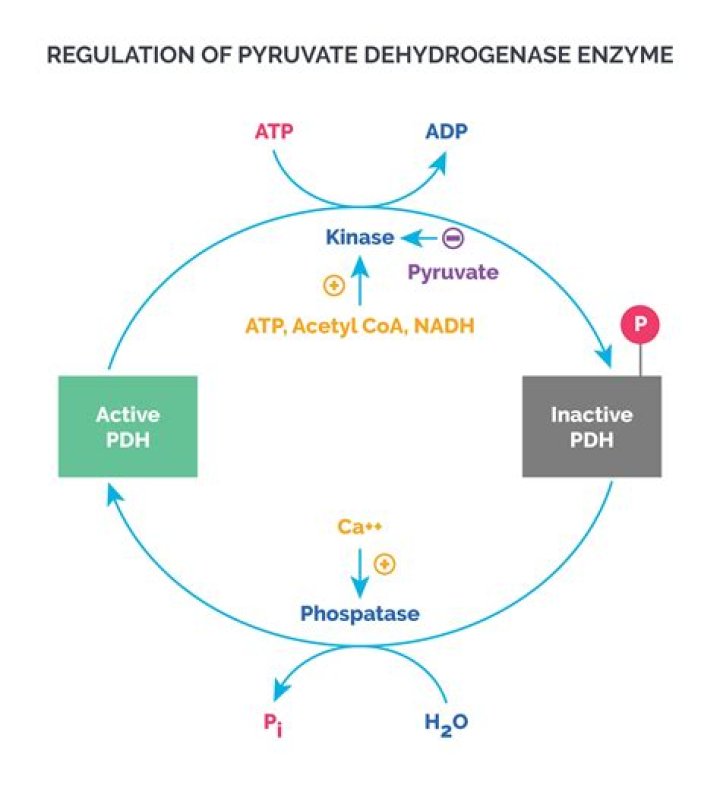
The enzyme that catalyzes the conversion reaction is called pyruvate dehydrogenase. ATP and NADH make this enzyme less active, while ADP makes it more active. So, more acetyl CoA is made when energy stores are low.
Does ATP stimulate pyruvate dehydrogenase?
Glycolysis and Pyruvate Oxidation The pyruvate dehydrogenase complex is regulated by covalent modification of the first enzyme, pyruvate dehydrogenase (PDH). Pyruvate dehydrogenase kinase inactivates PDH by phosphorylation with ATP (Fig. 6-5).
What stimulates the pyruvate dehydrogenase complex?
PDH kinase is stimulated by NADH and acetyl-CoA. It is inhibited by pyruvate. PDH phosphatase is stimulated by Ca++ and insulin.
What is the role of ATP ADP in PDH regulation?
This change in the relative concentration of ADP to ATP triggers the cell to slow down the electron transport chain. Electron Chain Transport: Levels of ADP and ATP affect the rate of electron transport through this type of chain transport.How does pyruvate affect ATP?
In the conversion of pyruvate to acetyl CoA, each pyruvate molecule loses one carbon atom with the release of carbon dioxide. During the breakdown of pyruvate, electrons are transferred to NAD+ to produce NADH, which will be used by the cell to produce ATP.
How many ATP are produced in pyruvate dehydrogenase?
Note that this process completely oxidizes 1 molecule of pyruvate, a 3 carbon organic acid, to 3 molecules of CO2. During this process, 4 molecules of NADH, 1 molecule of FADH2, and 1 molecule of GTP (or ATP) are produced.
Is pyruvate dehydrogenase inhibited by ATP?
ATP, acetyl CoA, and NADH negatively regulate (inhibit) pyruvate dehydrogenase, while ADP and pyruvate activate it.
What causes pyruvate dehydrogenase deficiency?
PDC deficiency is caused by genetic changes ( mutations or pathogenic variants) in any of the genes that provide instructions for the body to make the enzymes of the pyruvate dehydrogenase complex. Genes in which pathogenic variants are known to cause PDC deficiency include: PDHA1, PDHB, DLAT, PDHX, DLD, and PDP1.What is the role of pyruvate dehydrogenase PDH complex?
Pyruvate dehydrogenase complex (PDC) is a complex of three enzymes that converts pyruvate into acetyl-CoA by a process called pyruvate decarboxylation. … Pyruvate decarboxylation is also known as the “pyruvate dehydrogenase reaction” because it also involves the oxidation of pyruvate.
What is the name of the mechanism by which pyruvate dehydrogenase is inhibited by the end product of the biochemical pathway?The mechanisms that control PDC activity include end product inhibition by increased mitochondrial acetyl-CoA, NADH and ATP concentrations (which can also be generated by FA oxidation) and post-translational modification, namely its phosphorylation (inactivation) by a family of pyruvate dehydrogenase kinases (PDHKs 1–4 …
Article first time published onWhat produces pyruvate dehydrogenase?
Pyruvate dehydrogenase is an enzyme that catalyzes the reaction of pyruvate and a lipoamide to give the acetylated dihydrolipoamide and carbon dioxide. The conversion requires the coenzyme thiamine pyrophosphate.
Does pyruvate inhibit pyruvate dehydrogenase?
Pyruvate dehydrogenase (PDH) catalyzes an irreversible and no return metabolic step because its substrate pyruvate is gluconeogenic or anaplerotic, whereas its product acetyl-CoA is not [62–65]. … Pyruvate inhibits, whereas acetyl-CoA stimulates, PDK.
How does glucagon affect pyruvate dehydrogenase?
In the presence of 2.5 mM-Ca2+, glucagon caused an increase in NAD(P)H fluorescence in hepatocytes. This increase is taken to reflect an enhanced activity of mitochondrial dehydrogenases. … PMA alone had no effect on pyruvate kinase activity, and the phorbol ester did not prevent the inactivation caused by glucagon.
Is ATP used in pyruvate oxidation?
At the end of glycolysis, we have two pyruvate molecules that still contain lots of extractable energy. Pyruvate oxidation is the next step in capturing the remaining energy in the form of ATPstart text, A, T, P, end text, although no ATPstart text, A, T, P, end text is made directly during pyruvate oxidation.
How does pyruvate inhibit glycolysis?
Glycolysis Enzyme Inhibition Pyruvate kinase is inhibited by acetyl CoA, which is the molecule that pyruvate is converted to after glycolysis ends and before the Krebs cycle begins (in fact, acetyl CoA combines with oxaloacetate in the first step of the cycle to create citrate).
How much ATP will be produced by the complete oxidation of one pyruvate start with the pyruvate in the mitochondrial matrix?
So in total 15 ATPs are produced from one molecule of pyruvate. Further reading: Pyruvate.
Which of the following conditions will inhibit the pyruvate dehydrogenase complex?
The PDH complex is inhibited by high levels of NADH, so glucose is spared and acetyl CoA is used in the synthesis of fatty acids.
Does ADP regulate pyruvate dehydrogenase?
Pyruvate dehydrogenase activity is regulated additionally by pyruvate, inorganic phosphate, and ADP.
When ATP is in excess in cells acetyl CoA is used to produce?
Fatty acids are oxidized through fatty acid or β-oxidation into two-carbon acetyl CoA molecules, which can then enter the Krebs cycle to generate ATP. If excess acetyl CoA is created and overloads the capacity of the Krebs cycle, the acetyl CoA can be used to synthesize ketone bodies.
How many ATP are produced during the conversion of pyruvate to acetyl CoA?
The net gain from glycolysis is two molecules of ATP and two molecules of NADH. The conversion of pyruvate to acetyl CoA and its metabolism via the citric acid cycle yields two additional molecules of ATP, eight of NADH, and two of FADH2.
How many ATP does ATP synthase produce?
Function of ATP Synthase Each ATP synthase can produce about 100 molecules of ATP every second.
What are the components of the pyruvate dehydrogenase PDH )? And what is the function of each component?
PDH is a 9.5 MDa complex consisting of multiple copies of three enzymes: pyruvate dehydrogenase (E1), dihydrolipoamide transacetylase (E2) and dihydrolipoamide dehydrogenase (E3). An additional structural subunit, the E2/E3 binding protein, is necessary to support the interactions between the E2 and E3 subunits.
What role does pyruvate dehydrogenase play in metabolism?
One of the major enzymes responsible for metabolic flexibility in mammals is the pyruvate dehydrogenase complex (PDC), a mitochondrial multi-enzyme complex that catalyzes the oxidative decarboxylation of pyruvate [5]. … Thus, the PDC occupies a central position in cellular energy metabolism (Figure 1).
What is the main fate of pyruvate under anaerobic conditions in mammals?
Anaerobic use of Pyruvate The fate of pyruvate depends on the availability of oxygen. If oxygen is available, then pyruvate is shuttled into the mitochondria and continues through several more biochemical reactions called the “Citric Acid Cycle.” This is called aerobic metabolism.
Where does pyruvate dehydrogenase occur?
Where is the pyruvate dehydrogenase complex located? In eukaryotes, the pyruvate dehydrogenase complex, like the enzymes for citric acid cycle and oxidation of fatty acids, is located in the mitochondrion, where is associated with the surface of the inner membrane facing the matrix.
What causes high pyruvate levels?
An elevated lactate-to-pyruvate (L:P) ratio may indicate inherited disorders of the respiratory chain complex, tricarboxylic acid cycle disorders and pyruvate carboxylase deficiency. Respiratory chain defects usually result in L:P. ratios above 20.
What causes hyperammonemia in pyruvate carboxylase deficiency?
Hypoglycemia. Oxaloacetate deficiency limits gluconeogenesis. Note: Hypoglycemia is not a consistent finding despite the fact that PC is the first rate-limiting step in gluconeogenesis. Hyperammonemia results from poor ammonia disposal and decreased urea cycle function.
Why would amp stimulate cellular respiration and ATP inhibit it?
Why would AMP stimulate cell. respiration and ATP inhibit it? As ATP accumulates, inhibition of the enzyme slows down glycolysis and respiration as it is attempting to spare valuable organic molecules for other functions.
Why is ATP required for glycolysis?
Why is ATP required for glycolysis? ATP makes it easier to break apart glucose into two three-carbon molecules.
Why must PDH be tightly regulated?
In glucose metabolism, pyruvate dehydrogenase complex (PDC) mediates a major regulatory step, an irreversible reaction of oxidative decarboxylation of pyruvate to acetyl-CoA. Tight control of PDC is critical because it plays a key role in glucose disposal.
Which are the products of pyruvate dehydrogenase quizlet?
What are the products of PDH complex? For each molecule of pyruvate, you get one Acetyl CoA, one CO2 molecule, and one NADH.