Is the anode positive or negative in an electrochemical cell
Explanation: Reduction always occurs at the cathode, and oxidation always occurs at the anode. Since reduction is the addition of electrons, electrons must travel toward the site of reduction. In an electrolytic cell the negative charge is on the cathode, while the positive charge is on the anode.
Is cathode positive or negative in electrochemical cell?
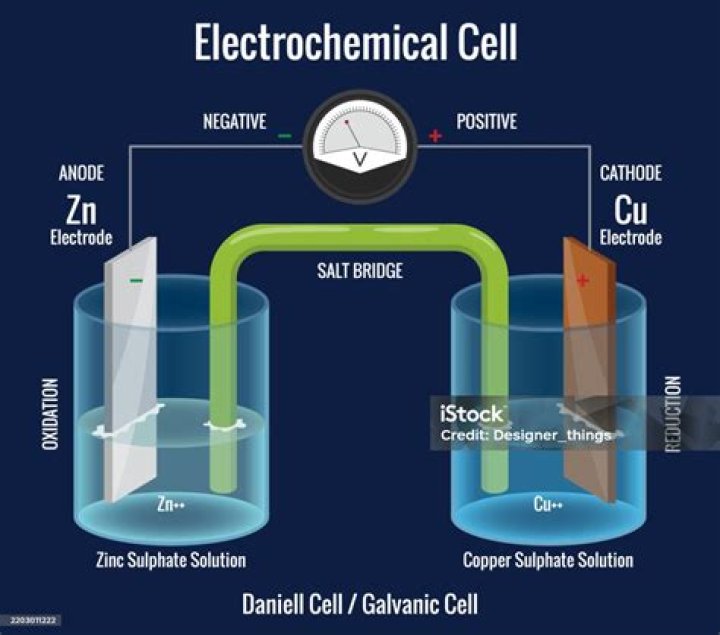
In a galvanic (voltaic) cell, the anode is considered negative and the cathode is considered positive. This seems reasonable as the anode is the source of electrons and cathode is where the electrons flow. However, in an electrolytic cell, the anode is taken to be positive while the cathode is now negative.
Which type of electrochemical cell has a negative anode?
In a galvanic (voltaic) cell, the anode is considered negative and the cathode is considered positive. This seems reasonable as the anode is the source of electrons and cathode is where the electrons flow. However, in an electrolytic cell, the anode is taken to be positive while the cathode is now negative.
What is the anode in an electrochemical cell?
In both kinds of electrochemical cells, the anode is the electrode at which the oxidation half-reaction occurs, and the cathode is the electrode at which the reduction half-reaction occurs. Table 1: Properties of Galvanic and Electrochemical Cells. Electrochemical cell (Galvanic Cell)Can an anode be negative?
In a battery or other source of direct current the anode is the negative terminal, but in a passive load it is the positive terminal. For example, in an electron tube electrons from the cathode travel across the tube toward the anode, and in an electroplating cell negative ions are deposited at the anode.
What is the anode and cathode in an electrochemical cell?
The anode is defined as the electrode where oxidation occurs. The cathode is the electrode where reduction takes place. … The voltaic cell uses two different metal electrodes, each in an electrolyte solution. The anode will undergo oxidation and the cathode will undergo reduction.
Does an anode have a positive charge?
In electronic vacuum devices such as a cathode ray tube, the anode is the positively charged electron collector. In a tube, the anode is a charged positive plate that collects the electrons emitted by the cathode through electric attraction.
Why is the anode negatively charged?
In a galvanic cell, electrons will move in to the anode. Since electrons carry a negative charge, then the anode is negatively charged. … It’s because the protons are attracted to the cathode, so it’s mainly positive, and therefore is positively charged.Why is the anode positive?
In an electrolytic cell(one used for electrolysis), the anode is positively charged. This is because the electrode connected to the positive terminal of the battery is where the negatively charged ions go to lose electrons, that is, to get oxidized.
Is anode positive or negative in electrophoresis?Explanation: Gel electrophoresis is a technique used to separate molecules based on size or charge. … In gel electrophoresis, the positive pole is called the anode and the negative pole is called the cathode; therefore, the charged particles will migrate to the respective nodes.
Article first time published onWhy anode is positive and cathode is negative in electrolytic cell?
1: An electrolytic cell. The battery pumps electrons away from the anode (making it positive) and into the cathode (making it negative). The positive anode attracts anions toward it, while the negative cathode attracts cations toward it. … Since the anode can accept electrons, oxidation occurs at that electrode.
Why anode is positively charged in electrolytic cell?
In an electrolytic cell(one used for electrolysis), the anode is positively charged. This is because the electrode connected to the positive terminal of the battery is where the negatively charged ions go to lose electrons, that is, to get oxidized.
Why is anode in a galvanic cell negative?
The anode is a negative electrode in the case of galvanic cells because when oxidation occurs, the electrons are left behind on the electrode itself. On the cathode, the positive ions are reduced to metal and this why this electrode is known as the positive electrode in a galvanic cell.
Which statement is true for an electrochemical cell?
Statement 1: In an electrochemical cell, the electrode that is the site of reduction is called the anode. Statement 2: Oxidation always occurs at the cathode.
Does a cathode have a positive charge?
The cathode is the electrode where electricity is given out or flows out of. The anode is usually the positive side. A cathode is a negative side. It acts as an electron donor.
What is the charge of cathode in an electrochemical cell?
– At cathode, reduction reactions occur and as reduction reaction requires electrons, the cathode will always have roughly no concentration of electrons on itself. So, it is positively charged.
Is the positive electrode the anode or cathode?
The Anode is the negative or reducing electrode that releases electrons to the external circuit and oxidizes during and electrochemical reaction. The Cathode is the positive or oxidizing electrode that acquires electrons from the external circuit and is reduced during the electrochemical reaction.
How can you tell which electrode is positive?
Usually, the black line is the COMMON and the red line is sometimes marked with a V. Attach the lines however you want to the electrochemical cell. If the voltage is positive then the red line is the anode (negative), and the black line is the cathode (positive).
Which ion has a positive charge?
Positively charged ions are called cations; negatively charged ions, anions.
Which side is positive in gel electrophoresis?
The bottom side of the gel is positive. DNA gel electrophoresis is used to separate DNA fragments based on size.
Which end is positive in gel electrophoresis?
In the electrophoresis gel, DNA samples are loaded in well close to the cathode (negative) end. Gel is porous, hence allows negatively charged DNA fragments to travel through, towards the anode (positive) end of gel.



