Kf Chemistry
What are KF and KR?
The equilibrium constant (K) for each reaction is the ratio of the direct reaction rate constant (Kf) to the reverse reaction rate constant (Kr). K = Kf / Kr. Kf and Kr remain different, but the respective speed remains the same due to the variation in concentration.
With this in mind, which speed constant is greater than KF or Kr?
The reaction C → D has a higher kf, a lower kr and a higher Kc (because it has the lowest activation energy for the forward direction). Remember that a large k (rate constant) means that the reaction is fast and therefore the reaction has a low activation energy.
What does a high KF value also mean?
When K is a large number, it means that the equilibrium concentration of the products is large. In this case the reaction proceeds as described on the right (which leads to an increase in the concentration of the products). If K is a small number, it means that the equilibrium concentration of the reactants is large.
People also ask: What is KF in chemical equilibrium?
Chemical reactions, reversible or irreversible, follow chemical kinetics. Hence, we can better understand the equilibrium constant K if we use the term chemical kinetics. Let kf be the reaction rate and kr the reverse reaction rate, as shown below.
What is KF in solubility?
Many metal sulfides are insoluble in water but dissolve in acidic solutions. The formation of complex ions or the stability constant Kf of a complex ion is the equilibrium constant for the formation of the complex ion from the aqueous metal ion and ligands. Does this mean that there are 1.
5 106 times more product molecules in equilibrium than there are reactant molecules?
The magnitude of the equilibrium constant K depends on the relative rate constants, not on the reaction rate. The statement is vaguely correct. If Ke = 1.5 * 106, the arithmetic product of the concentrations of all product molecules is 1.5 * 106 times the product of the concentrations of all reactant molecules.
Is KF an acid or a base?
KF is a salt composed of a strong base, KOH, and a weak acid, HF. When salt is added to water, it dissociates into a K + ion and an F ion. The F ion, the conjugate base of the weak acid, reacts with water, absorbs a hydrogen atom from the water and forms hydroxide .
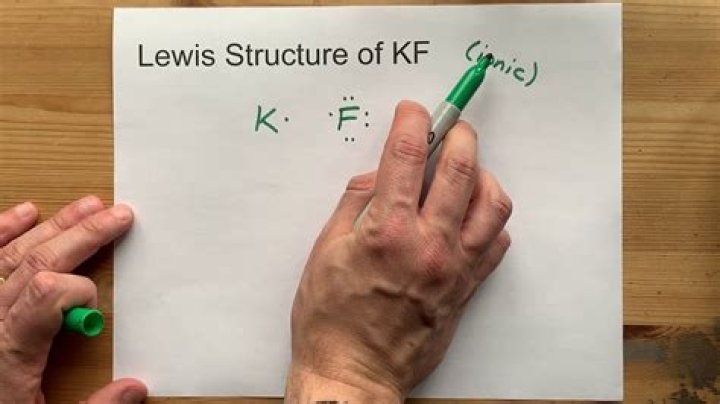
What is the formula for KF?
To calculate the freezing point depression constant, or Kf, you need the equation: Delta Tf = Kfcm where cm is the molar concentration of the solution. Write what you know. Since Kf is a constant or the same number, it is often given in a graph or table in chemistry textbooks.
What is the KF value?
Kf is the constant molar depression of the freezing point for the solvent (1.86 ° C / m for water). m = molality = ■■■■■ of solute per kilogram of solvent.
Is KF soluble in water?
What is K in chemistry?
K is very important in chemistry. In inorganic chemistry, K (capital letter) is used to denote the element potassium, which means that K is the symbol for potassium. In physical chemistry, k is used to describe the equilibrium constant or the solubility constant.
(Keq = equilibrium constant Ksp = solubility constant)
What is the mathematical relationship between KF and KD?
the correct relationship between kf and kd is kf = kd, kf = 1 / kd, kf = 1 / kd squared, kf = square root of kd, all Brainly.in.
What am I in a freezing point?
Freezing point depression is the lowering of the freezing point of a solvent by adding a non-volatile solvent. Examples are the salt in the water, the alcohol in the water, or a mixture of two solids such as the impurities in a fine powdered drug.
What is the steady gradual training?
A stability constant (formation constant, bond constant) is an equilibrium constant for the formation of a complex in solution. It is a measure of the strength of the interaction between the reactants that make up the complex.
Q changes with temperature?
What does KEQ 1 mean?
If Keq = 1, then the equilibrium position is in the middle, the amount of product is approximately equal to the amount of starting materials in equilibrium. If Keq is much smaller than 1 (e.g. at Keq = 103), then the equilibrium position on the left is that several reactants are in equilibrium.
What affects the KEQ?
An equilibrium system is influenced by the following factors:
What does it mean if k is greater than 1?
When K is greater than 1, the mixture generally contains products. K <1. If K is less than 1, the mixture usually contains reagents. K = 1. When K is approximately equal to 1, the intermediate reaction balances, which means that the quantities of products and reactants are approximately the same.
Does a large equilibrium constant mean that products or reactants are preferred?
The value of K indicates the equilibrium relationship between products and reactants. Both the starting materials and the products are in an equilibrium mixture.
Large products are preferred K> 1
Does a large equilibrium constant mean that the reaction must be fast?
Why does K change with temperature?
Since the value of K decreases with increasing temperature, it is an exothermic reaction. 4. However, if the temperature is raised, the system can absorb energy and thus promote an endothermic reaction, the equilibrium shifts to the left.
Can the equilibrium constant be zero?
Kf Chemistry
![Complex Ion Equilibria - Stepwise Formation Constant Kf & Ksp Molar Solubility Problems]()