What happens when formaldehyde reacts with ammonia
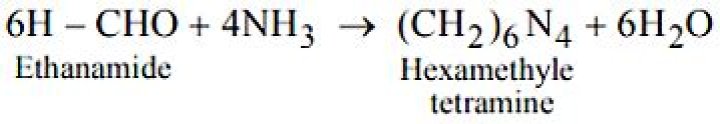
It is well known that formaldehyde reacts with ammonia to form hexamethylenetetramine or hexamine (VI) in a good yield.
What happens formaldehyde reacts with ammonia?
Ammonia reacts with formaldehyde to form a condensation product, hexamethylene tetramine, commonly named as Urotropine.
Does ammonia neutralize formaldehyde?
… After this, neutralization takes place by heating up ammonia bicarbonate or ammonium carbonate to generate an ammonia vapor. This ammonia vapor then neutralizes the formaldehyde gas and creates a relatively safe byproduct called methenamine (Luftman, 2005) .
What is the action of ammonia on a formaldehyde?
Explanation: When formaldehyde reacts with ammonia, it forms methyleneimine, ethyl alcohol and hexamethylenetetramine. In general, aliphatic aldehydes react with ammonia and form aldehyde ammonias.How does ammonia react with formaldehyde and acetaldehyde?
Formaldehyde an aliphatic aldehyde reacts with ammonia in slightly alkaline or neutral medium and forms amino methanol as the initial product. Later amino methanol undergoes reaction with formaldehyde and forms a methylene derivative as the product.
What happens when formaldehyde is treated with NaOH?
When treatment of formaldehyde with conc. NaOH solution, methanol and sodium formate are formed. An organic compound contains 69.77% carbon, 11.63% hydrogen, and oxygen residue.
Which product is formed when formaldehyde reacts with concentrated ammonia?
It is well known that formaldehyde reacts with ammonia to form hexamethylenetetramine or hexamine (VI) in a good yield.
What is the formula of formaldehyde?
The chemical formula of formaldehyde is CH2O: it has one carbon (C) atom, two hydrogen (H) atoms and one oxygen (O) atom.Where is formaldehyde used?
Formaldehyde is a strong-smelling, colorless gas used in making building materials and many household products. It is used in pressed-wood products, such as particleboard, plywood, and fiberboard; glues and adhesives; permanent-press fabrics; paper product coatings; and certain insulation materials.
What neutralizes formaldehyde?Formaldehyde polymerization by use of urea is a proven method of neutralizing formaldehyde. The reaction of formaldehyde with urea and acid will neutralize the formaldehyde.
Article first time published onWhat neutralizes formaldehyde smell?
Removal of Lingering Odors If it still smells like formaldehyde in house, sprinkle baking soda over the upholstery to remove lingering odors. Sprinkle the baking soda liberally over the fabric and allow it to sit for about an hour, then vacuum it away with a vacuum cleaner containing a clean air or HEPA filter.
What neutralizes formaldehyde Hcho?
sodium hydroxide (NaOH) reacts with formaldehyde (HCHO), the products formed is/are sodium formate (sodium-formiate, HCOONa , and methanol (CH3OH).
What happens when acetone and formaldehyde is seperately treated with ammonia?
Aldehydes, Ketones and Carboxylic Acids When formaldehyde is treated with ammonia, it forms urotropine.
What happens when formaldehyde reacts with hydrazine?
In summary, the data support, in part, the origianl hypothesis, that hydrazine reacts with endogenous formaldehyde in the liver cell to form formaldehyde hydrazone which is oxidized to diazomethane.
What happens when formaldehyde is heated?
At room temperature, formaldehyde is a colorless, flammable gas that has a distinct, pungent smell. … Vapor from formalin solution is flammable and an explosion hazard when exposed to flame or heat. Skin and eye irritant. Confirmed carcinogen.
When formaldehyde is treated with ammonia a crystalline compound urotropine?
When formaldehyde reacts with ammonia, a well known urinary antiseptic urotropine (also called hexamethylenetetramine) is obtained. Hence Urotropine is formed by the action of Ammonia on Formaldehyde. Hence, Option B is the correct answer.
What is the use of urotropine?
It is also called Methenamine or Hexamethylenetetramine or Urotropin. It acts as an anti-infective agent which is most commonly used to treat urinary tract infections.
Which of the following on reaction with ammonia gives urinary antiseptic compound?
When formaldehyde reacts with ammonia, a well known urinary antiseptic urotropine (also called hexamethylene tetramine) is obtained.
What happens when formaldehyde is treated with concentrated KOH solution?
Formaldehyde when treated with KOH gives methanol and potassium formate.
What is the reaction between formaldehyde and concentrated base?
4) Crossed Cannizzaro reaction: When a mixture of formaldehyde and a non enolizable aldehyde is treated with a strong base, the latter is preferentially reduced to alcohol while formaldehyde is oxidized to formic acid. This variant is known as cross Cannizzaro reaction.
When two molecules of formaldehyde reacts in the presence of base to produce methyl alcohol and sodium formate The reaction is known as?
The Cannizzaro reaction, named after its discoverer Stanislao Cannizzaro, is a chemical reaction which involves the base-induced disproportionation of two molecules of a non-enolizable aldehyde to give a primary alcohol and a carboxylic acid.
Is formalin the same as formaldehyde?
Formalin is an alternative name for an aqueous solution of formaldehyde, but the latter name is preferred, since formalin is also used as a brand name in some countries. Free formaldehyde is used in cosmetics, especially in hair shampoos, and in many disinfectants and antiseptics.
Why is formaldehyde used in nail polish?
For example, toluene sulfonamide/formaldehyde resin (TSFR) is used in some nail polishes to make the coating tough and resilient. TSFR also helps the polish adhere to the nail, adds gloss and helps the product flow well when applied. There is evidence that some people may become allergic to TSFR.
Why is formalin added to milk?
formalin binds with protein and affects biological and functional properties of proteins. when added more than 40 % level it increases the acidity of milk,reducs Fat % and, Protein but increases the Viscosity of milk. , but more importantly, this inhibits the bacteriological growth in milk.
What is the pH of formaldehyde?
Like all aldehydes, formaldehyde absorbs oxygen comparatively readily even from the air and is thereby oxidised to formic acid. Solutions of formaldehyde attain fairly rapidly and maintain a pH of 3.5 or even of 3.
Do humans produce formaldehyde?
Humans produce about 1.5 ounces of formaldehyde a day as a normal part of our metabolism. Inhaled formaldehyde is rapidly metabolized and ultimately converted to carbon dioxide and exhaled. Formaldehyde does not accumulate in the body.
What is formalin BYJU's?
To recall about formaldehyde, it is also known as methanal or formalin, is an organic compound which occurs naturally. … It is the simplest aldehyde and is colorless.
Does baking soda remove formaldehyde?
Formaldehyde in clothing also makes it more flammable, and can cause contact dermatitis. Washing will remove the chemical treatment. Baking soda adsorbs (yes, that’s spelled right, it is a chemical process, no a physical one) some of it, but does not neutralize it. … Formaldehyde is readily soluble in water.
Can formaldehyde be stored in plastic?
Any container made of inert material is OK. Any plastic is OK, and is not essential.
Will an air purifier remove formaldehyde?
Air purifiers with activated carbon filters can eliminate formaldehyde and hundreds of other chemicals from the ambient air. Activated carbon has a high-efficiency rating when it comes to removing formaldehyde.
Why do old people smell like formaldehyde?
Starting at about age 40, human bodies begin to subtly change the way that omega-7 unsaturated fatty acids on the skin are degraded. … When lipid acid is oxidized, the chemical compound nonenal is produced, giving off the “old people smell” that many of us are familiar with.