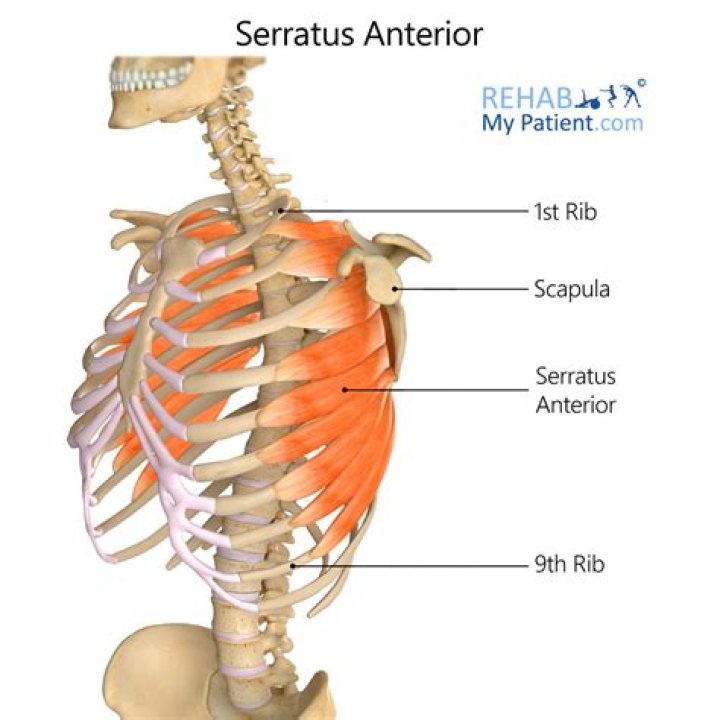
What will be formed after oxidation reaction of 2 degree alcohol with chromic acid
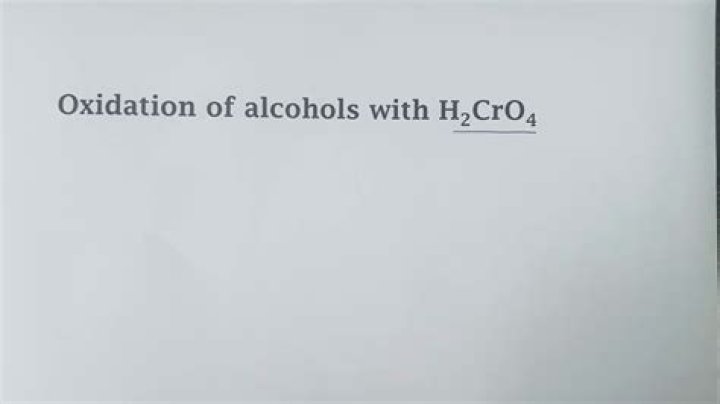
Chromic acid oxidizes primary alcohols to carboxylic acids, and it oxidizes secondary alcohols to ketones.
What will be formed after oxidation of 2 degree alcohol with chromic acid?
Chromic acid is most commonly used to oxidize 2o alcohols to ketones.
What is the product of the oxidation of ethanol with chromic acid?
In the first step of the reaction, ethanol reacts with chromic acid to form what’s called a chromate ester. Once the chromate ester is formed, it then breaks down by reaction with a base to form an aldehyde.
What does cro3 do to a secondary alcohol?
A primary alcohol is oxidized to an aldehyde or all the way to a carboxylic acid, while a secondary alcohol to a ketone. The mechanism begins with the reaction of CrO3 with acid (often H2SO4) to form chromic acid or dichromic acid in more concentrated solutions.Can chromic acid oxidize a secondary alcohol?
Alcohol Oxidation Reactions with Chromic Acid Chromic Acid is the stronger of the two oxidizing agents. It will carry out both steps of oxidation of a primary alcohol producing a carboxylic acid as well as oxidizing secondary alcohols to ketones. It will also oxidize an aldehyde to a carboxylic acid.
How is chromic acid formed?
Chromic acid is formed when chromium trioxide reacts with water. Chromium trioxide is crystalline, light red or brown in colour and is deliquescent and fully soluble in water. Chromic acid is a very weak acid and its salts can be dissociated even by acetic acid.
What is the product of the oxidation of 2 Pentanol?
Overview – 2- Pentanol will be oxidized using hypochlorous acid (HOCl) to form 2-pentanone (eq 1). The HOCl is generated in situ from the reaction of acetic acid with aqueous sodium hypochlorite (Clorox).
What is a chromic acid test?
The chromic acid test uses the Jones reactant to oxidize aldehydes and alcohols and reduce the chromic acid, resulting in a color change. It is able to identify aldehydes, primary alcohol, and secondary alcohol.What is chromic acid oxidation?
Chromic acid (H2CrO4) oxidizes alcohols in aqueous solutions of sodium dichromate. It reacts with alcohols to form a chromic ester in which the alcohol oxygen atom bridges the carbon and chromium atoms. Thus, the ester forms by nucleophilic attack of the alcohol’s oxygen atom on the chromium atom.
What is the use of chromic acid?Chromic Acid (Dichromic Acid, Chromium Trioxide) is most known for its use in the metal finishing (intermediate in chromium plating) industry. Other common uses are as a wood preservative, production of plastic products, ceramic glazes and to clean laboratory glassware.
Article first time published onWhen phenol is treated with chromic acid then the product formed is?
Oxidation of phenol with chromic acid gives benzoquinone.
Which compound is oxidized to a ketone by chromic acid?
secondary alcohols are oxidized to ketones while the Cr+6 ion in the chromic acid is reduced to Cr+3.
Which alcohol will not be oxidized by chromic acid?
Ketones are not oxidized by chromic acid, so the reaction stops at the ketone stage. In contrast, primary alcohols are oxidized by chromic acid first to aldehydes, then straight on to carboxylic acids.
How can you distinguish between a primary and secondary alcohol using chromic acid?
Alcohols are organic molecules containing a hydroxyl functional group connected to an alkyl or aryl group (ROH). If the hydroxyl carbon only has a single R group, it is known as primary alcohol. If it has two R groups, it is a secondary alcohol, and if it has three R groups, it is a tertiary alcohol.
How is chromic acid formed by the reaction of potassium dichromate and sulfuric acid?
Chromic acid is prepared by treatment of sodium or potassium dichromate with aquesous sulfuric acid as shown in Equation 1. … Pyridinium chlorochromate is made by mixing chromium trioxide with pyridine and hydrochloric acid as indicated in Equation 3. The oxidizing component of PCC is the chlorochromate anion, CrClO3-.
Does chromic acid react with carboxylic acid?
Chromic acid, H2CrO4, is a strong acid and a reagent for oxidizing alcohols to ketones and carboxylic acids.
How can we reduce aldehydes?
The reduction of aldehydes and ketones by sodium tetrahydridoborate. Sodium tetrahydridoborate (previously known as sodium borohydride) has the formula NaBH4, and contains the BH4- ion. That ion acts as the reducing agent.
What is the reduction of butanone?
2-butanone is a 4-carbon chain with a double bond between an oxygen and carbon 2. The reduction turns the double bond with oxygen into a single bond to a hydroxy group. This makes the product 2-butanol.
What is oxidation number of Cr in chromic acid?
Chromic acid features chromium in an oxidation state of +6 (or VI). It is a strong and corrosive oxidising agent.
What type of chemical is chromic acid?
Chromic acid is a chromium oxoacid. It has a role as an oxidising agent.
How do you make chromic acid from chromium trioxide?
The chromic acid oxidizing reagent is prepared by dissolving 67 g. of chromium trioxide in 125 ml. of distilled water. To this solution is added 58 ml.
Is chromic acid a primary alcohol?
It is a strong oxidant, and it oxidizes the alcohol as far as possible without breaking carbon-carbon bonds. Chromic acid oxidizes primary alcohols to carboxylic acids, and it oxidizes secondary alcohols to ketones.
How is chromic acid reagent prepared?
Chromic acid is a commonly used glassware cleaning reagent. It is prepared in a one liter container by dissolving 60 grams of potassium dichromate in approximately 150 mls of warm distilled water and then slowly adding concentrated sulfuric acid to produce a total volume of one liter Chromic Acid solution.
What results are expected when the chromic acid test is done on a?
A change in the solution’s color from red orange (chromic acid) to blue green (Cr(III)) ion indicates a positive result. Alcohols (except tertiary) and aldehydes give a positive result since there is an available proton from the carbon which can be removed during the reaction.
Is chromic acid inorganic?
Chromic acid and chromium trioxide are inorganic compounds.
What happens when phenol is oxidized by chromic acid?
Textbook solution. Oxidation of phenols gives benzoquinone. … When phenol is oxidized using chromic acid in presence of sulphuric acid and water it forms benzoquinone.
What are the products formed when phenol oxidised?
b) Oxidation of phenol: Phenol undergo oxidation with chromicacid and forms a conjugated diketone known as benzoquinone.
What is oxidation product of phenol?
Oxidation of phenol gives p-benzoquinone.
Which alcohol can be oxidized to a ketone?
Primary alcohols can be oxidized to form aldehydes and carboxylic acids; secondary alcohols can be oxidized to give ketones. Tertiary alcohols, in contrast, cannot be oxidized without breaking the molecule’s C–C bonds.
Which one of the following alcohols will be oxidized to a ketone?
Primary alcohol- They are oxidised to either aldehydes or carboxylic acids depending upon the reaction condition. For the carboxylic acid formation, alcohol is first oxidised into an aldehyde which is then further oxidised into carboxylic acid. Secondary alcohol– They are oxidised to ketones.
Why are secondary alcohols oxidized to ketones?
Where a secondary alcohol is oxidised, it is converted to a ketone. The hydrogen from the hydroxyl group is lost along with the hydrogen bonded to the carbon attached to oxygen. The remaining oxygen then forms double bonds with the carbon. This leaves a ketone, as R1–COR2.